12-Jan-2021
Transcription and replication of alphaviruses such as chikungunya take place in a coordinated fashion in virus-induced membranous organelles called spherules within host cells. The formation of these viral niches facilitates viral RNA synthesis and protects viral RNA from the host.
Recent work at the AFMB in Marseille made in collaboration with the Cryo-EM facility in the Spanish Instruct Centre CSIC in Madrid reveals the structure of nsP1, a non-structural capping enzyme from alphaviruses viral replication complex, that plays a crucial role in formation of these spherules, virus replication and release of capped viral RNA within host cells. The characterisation of this viral replication complex is very important to understand how this type of virus interacts with the host and could open the door to the generation of a new class of antiviral agents.
In their study, Rhian Jones and colleagues expressed nsP1 of chikungunya virus in E.coli and insect cells and used single particle Cryo-EM microscopy to determine the structure of the active nsP1 protein complex. The structures were determined to a resolution of 2.6A.
The Cryo-EM structure shown in Figure 1 reveals that in its active form, nsP1 forms ring complexes composed of 12 subunits that associate with the cytoplasmic membrane.
This ring complex is composed of a capping domain: the crown, the waist domain that forms a pore that would allow the passage of small proteins and other particles and the skirt that associates with the membrane
The structure shows that the membrane association and the oligomerization itself allows the activation of the nsP1 capping enzyme by stabilizing the conformation of the capping domain and creating a catalytic pocket composed of the GTP-binding domain and the SAM-binding domain.
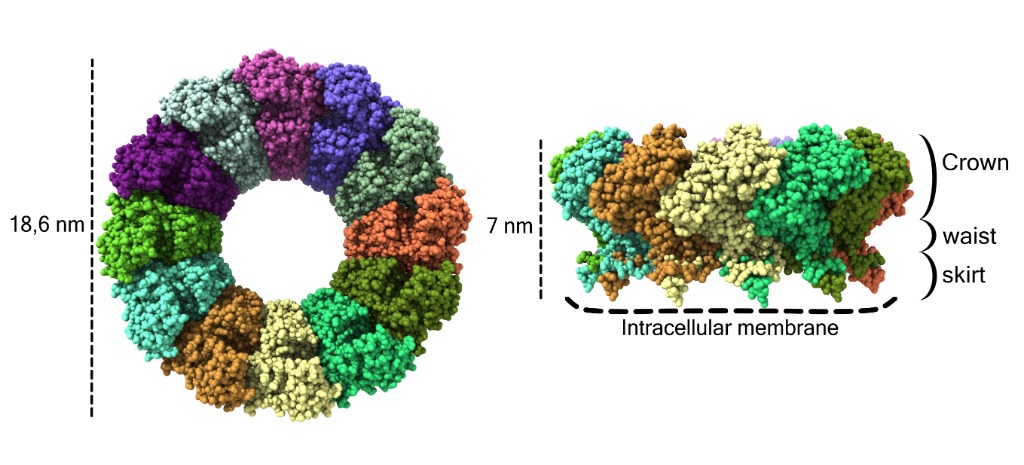
Figure1: Cryo-EM structure of the spherule–pore complex of CHIKV nsP1. Atomic structure of the pores of the Chikungunya nsP1 virus with the different monomers shown in different colors. Each atom is represented as a sphere. On the left side of the image the pore is shown from the cytoplasmic view and on the right from a side view. The dimensions of the pore are indicated on the left, the crown, waist and skirt regions are indicated on the right and the contact sites with the membrane are indicated at the bottom by dashed lines.
Tomographic reconstruction of viral spherule necks, that connect the viral niche with the external cytosol also shows strong similarity in size and structure with the nsP1 ring complex.
These results indicate that nsP1 together with the other proteins from the viral replication complex play an essential role in the formation of virus-induced organelles within host cells and are likely creating a gate that allows the combined capping and release of viral RNA in the external cytosol within host cells.
Read the full article: Jones, R., Bragagnolo, G., Arranz, R. et al. Capping pores of alphavirus nsP1 gate membranous viral replication factories. Nature (2020). https://doi.org/10.1038/s41586-020-3036-8
Find out more about CryoEM facilities at Instruct-ERIC.