29-Jan-2021
In the past year, researchers around the world have worked to develop therapies and vaccines against the SARS-CoV-2 virus responsible for the COVID-19 pandemic. One of the main targets for vaccine development is the trimeric spike (S) protein. S is a large viral surface protein crucial for the viral life cycle that mediates both the interaction and the fusion of the SARS-CoV-2 virus with host cells.
Before fusing with host cells, the receptor-binding domain (RBD) of the S protein alternates between an open state and a closed state. Previous studies have shown that antibodies that recognise the RBD in a closed conformation have a higher neutralising potency, making the pre-fusion S-closed trimer an immunogen of great interest.
One major limitation in the development of vaccines or serologic tests targeting the S protein comes from the intrinsic instability of this trimer which makes the isolation of stable S-closed proteins in a pre-fusion state challenging.
To tackle this problem, a team from Janssen Vaccines & Prevention B.V collaborated with the Netherlands Center for Electron Nanoscopy (NeCEN) part of Instruct Centre NL to create and characterise new stable variants of the S trimer.
A series of non-exposed single mutations selected computationally were first introduced in the S protein and the effect of these mutations on protein expression, stability or trimer closure was assessed. Subsequently, a new set of protein S variants was produced based on the combination of mutations that had a positive effect on these three factors.
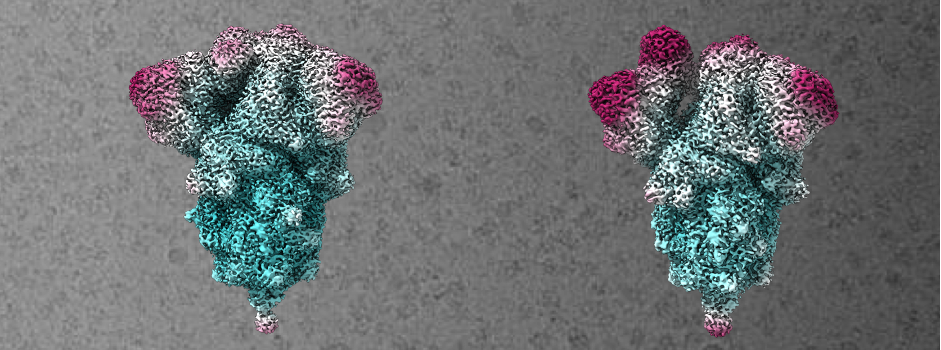
Figure 1: Cryo-EM structure of the most abundant trimer class of S-closed-Fd-a closed S protein trimer on the left (PDB accession code: 7A4N) and the less representative open trimer class on the right (PDB accession code: 7AD1). Colored by local resolution cyan being the highest resolution and purple the lowest.
Using this method, a stable S variant was produced that displayed 6.4 times higher expression than the parental protein construct, high thermal and freeze–thaw stability and antigenic characteristics expected for S trimers in a closed conformation. It was confirmed using Cryo-EM that the majority of these variants adopt a closed conformation (the 3 RBD of the trimers are down). The structure of this S-closed variant was resolved at 2.8 Å resolution using Cryo-EM (figure 1) which confirmed that the majority of these new S variants retain the pre-fusion conformation.
The production of this new stable Spike (S) variant could help tremendously in the development of more potent vaccine immunogens and may lead to considerable advances in the development of vaccines or serological diagnostics.
Read full text of the article Juraszek, J., Rutten, L., Blokland, S. et al. Stabilizing the closed SARS-CoV-2 spike trimer.Nat Commun 12, 244 (2021). https://doi.org/10.1038/s41467-020-20321-x
Read more about CryoEM at Instruct-ERIC.